Difference between revisions of "EMSegmenter-Tasks:MRI-Human-Brain-Parcellation"
| Line 53: | Line 53: | ||
The SUBGM and SUBWM were excluded for the studies done by Nakamura et al, 2007 but were added back to the manually segmented scans and were used in generating the atlases used for our segmentation purposes. | The SUBGM and SUBWM were excluded for the studies done by Nakamura et al, 2007 but were added back to the manually segmented scans and were used in generating the atlases used for our segmentation purposes. | ||
| + | |||
| + | Over multiple experiments it was found that the segmentation works best when WM atlases are given as those closest to a particular GM region as opposed to providing a single WM atlas. | ||
=Atlas= | =Atlas= | ||
Revision as of 17:01, 15 December 2010
Home < EMSegmenter-Tasks:MRI-Human-Brain-ParcellationReturn to EMSegmenter Task Overview Page
Contents
Description
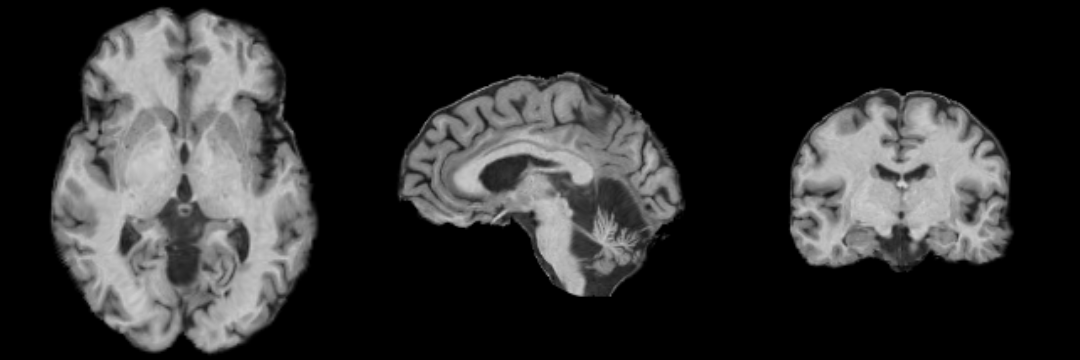
MRI Human Head pipeline for a finer-grained parcellation
The pipeline consist of the following steps:
- Step 1: Perform image inhomogeneity correction of the MRI scan via N4ITKBiasFieldCorrection (Tustison et al 2010)
- Step 2: Register the atlas to the MRI scan via BRAINSFit (Johnson et al 2007)
- Step 3: Compute the intensity distributions for each structure
Compute intensity distribution (mean and variance) for each label by automatically sampling from the MR scan. The sampling for a specific label is constrained to the region that consists of voxels with high probability (top 95%) of being assigned to the label according to the aligned atlas.
- Step 4: Automatically segment the MRI scan into the structures of interest using EM Algorithm (Pohl et al, 2007)
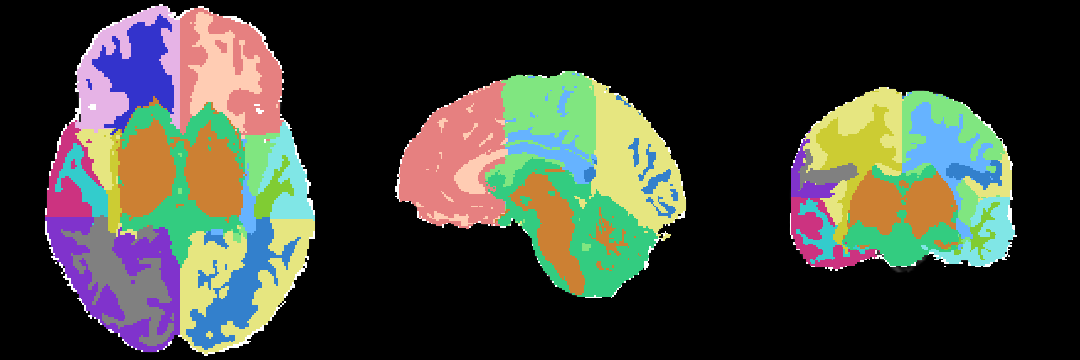
Anatomical Tree
The anatomical tree represents the structures to be segmented. Node labels displayed below contain a human readable structure name and in parentheses the internally used structure name.
- Root
- background (BG)
- grey matter (GM)
- left grey matter (LTGM)
- left grey matter - region 1 (LTGM1)
- left grey matter - region 2 (LTGM2)
- left grey matter - region 3 (LTGM3)
- left grey matter - region 4 (LTGM4)
- right grey matter (RTGM)
- right grey matter - region 1 (RTGM1)
- right grey matter - region 2 (RTGM2)
- right grey matter - region 3 (RTGM3)
- right grey matter - region 4 (RTGM4)
- subcortical grey matter (SUBGM)
- left grey matter (LTGM)
- white matter (WM)
- left white matter (LTWM)
- left white matter - region 1 (LTWM1)
- left white matter - region 2 (LTWM2)
- left white matter - region 3 (LTWM3)
- left white matter - region 4 (LTWM4)
- right white matter (RTWM)
- right white matter - region 1 (RTWM1)
- right white matter - region 2 (RTWM2)
- right white matter - region 3 (RTWM3)
- right white matter - region 4 (RTWM4)
- subcortical white matter (SUBWM)
- left white matter (LTWM)
- cerebrospinal fluid (CSF)
Description of manually segmented data used to generate the atlases
1.5T data that were previously manually segmented were used to generate the atlases. A brief description of the criteria used to segment the scans is given here, for more details the reader is referred to (Nakamura et al, 2007). EM algorithm (Pohl et al, 2007) was used to initially segment the scans into Gray Matter (GM), White Matter (WM) and Cerebro-Spinal Fluid (CSF). Manual editing then separated the Neo-cortical Gray Matter (NCGM) and its lobar parcellation, Cerebral WM, Sulcal CSF and Lateral Ventricles (LV).
From Nakamura et al, 2007: NCGM was manually parcellated into three lobar ROI, frontal, temporal, and parieto-occipital. This parcellation mainly used sulcal boundaries because these are more faithful to brain anatomy than a purely geometric parcellation. The frontal lobe was separated from the parieto-occipital lobe by the central sulcus on the convexity, a boundary that is constant, easily identifiable, and traceable with little interindividual variation (Ono et al, 1990). The central sulcus was initially traced on the axial plane, and subsequently these trace lines were used on the coronal plane to separate frontal and parietal lobes. For frontal GM on the medial wall, the posterior terminus was the most posterior coronal slice containing corpus callosum. The frontal lobe was clearly separated from the temporal lobe by the Sylvian fissure and circular insular sulcus. The posterior temporal lobe terminus was geometrically defined as the most posterior coronal slice where the fornix could be clearly seen along the lateral ventricles, as in the previous studies (Hirayasu et al, 2000). Occasionally, especially in the right hemisphere, the Sylvian fissure steeply ascended posteriorly through the parieto-occipital region. In this case, the superior boundary separating the temporal lobe from the parieto-occipital lobe was defined as the most superior axial slice where Heschl's gyrus could be seen. The parieto-occipital lobe was automatically defined by its contiguous boundaries with the frontal and temporal lobes.
The work done in Nakamura et al, 2007 does not differentiate between the pre-frontal and frontal lobe. But in our study a further parcellation was done between the two. The structural scans were segmented using the first slice in which the temporal stem was evident. The temporal stem is a piece of white matter connecting the temporal lobe to the external capsule/deep brain grey matter in coronal view. It lies just above the limen insula, just lateral to the anterior perforated substance. This is why the labels for left and right prefrontal lobe (PFL) are not in the same coronal slice in the volume you provided. Whatever lay in the first slice of the temporal stem, as well as posterior to the temporal stem (but still ahead of the precentral sulcus) was defined as frontal lobe proper (FL) (from one of the tracers Usman Khan).
The SUBGM and SUBWM were excluded for the studies done by Nakamura et al, 2007 but were added back to the manually segmented scans and were used in generating the atlases used for our segmentation purposes.
Over multiple experiments it was found that the segmentation works best when WM atlases are given as those closest to a particular GM region as opposed to providing a single WM atlas.
Atlas
The atlas has been created by Ryan Eckbo and Padmapriya Srinivasan and Sylvain Bouix from (PNL-BWH)
Image Dimension = 256 x 256 x 220
Image Spacing = 0.9375 x 0.9375 x 0.9375
Briefly, the steps followed for creating the atlases are described here:
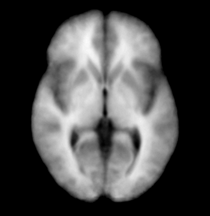
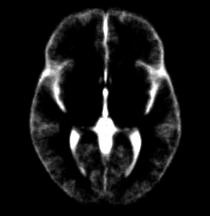
For each case from previous manually segmented 1.5T scans (Nakamura et al, 2007), binary label maps for the 20 leaves (BG,ltgm1-ltgm4, rtgm1-rtgm4, subgm, ltwm1-ltwm4, rtwm1-rtwm4, subwm and CSF) to be segmented were obtained. Initially, all these label maps were respectively affine transformed to a pre-determined case. A diffeomorphic registration algorithm (Vercauteren et al, 2008) was applied and this produced warps of all the regions for all the cases. The warps were applied to the original images and all the images for respective regions are aligned in a final atlas space. Probability maps are then determined by averaging the images for respective regions i.e. BG, ltgm1, ltgm2, CSF etc.
Weighted combined atlases: Left and right classification of the brain typically relies on the atlases since the intensities are similar. Thus if an area on the left side say ltgm1 is not covered adequately by the atlas, the segmentation algorithm puts the next region with the highest probability in the ltgm1 region. Therefore, in order to get an accurate segmentation and to prevent left and right mis-classification, the atlases were weighted and combined for each leaf. For example, for ltgm1 the ltgm1 atlas was weighted by 0.9 and the ltwm1 atlas was weighted by 0.1 and then summed up. Thus in this way, for ltgm1 the atlas covers even the white matter region except that the probability of finding grey matter is lower. The opposite if done for ltwm1 i.e. the ltwm1 atlas is weighted by 0.9 and ltgm1 is weighted by 0.1 and then summed up. These weighted combined atlases provide a much accurate segmentation.
|
|
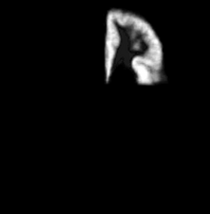
|
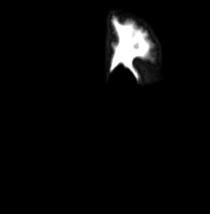
|
| Template (T1) | CSF | Left GM1 (LTGM1) | Left WM1 (LTWM1) |
Result
Collaborators
Padmapriya Srinivasan and Sylvain Bouix (PNL-BWH)
Acknowledgment
The construction of the pipeline was supported by funding from NIH NCRR 2P41RR013218 Supplement.
Citations
- Tustison NJ, Avants BB, Cook PA, Zheng Y, Egan A, Yushkevich PA, Gee JC N4ITK: Improved N3 Bias Correction, IEEE Trans Med Imag, 2010
- Pohl K, Bouix S, Nakamura M, Rohlfing T, McCarley R, Kikinis R, Grimson W, Shenton M, Wells W. A Hierarchical Algorithm for MR Brain Image Parcellation. IEEE Transactions on Medical Imaging. 2007 Sept;26(9):1201-1212.
- S. Warfield, J. Rexilius, P. Huppi, T. Inder, E. Miller, W. Wells, G. Zientara, F. Jolesz, and R. Kikinis, “A binary entropy measure to assess nonrigid registration algorithms,” in MICCAI, LNCS, pp. 266–274, Springer, October 2001.
- Johnson H.J., Harris G., Williams K. BRAINSFit: Mutual Information Registrations of Whole-Brain 3D Images, Using the Insight Toolkit, The Insight Journal, July 2007
- M. Nakamura, D. F. Salisbury, Y. Hirayasu, S. Bouix, K. Pohl, T. Yoshida, M. Koo, M. Koo, R. McCarley. Neocortical Gray Matter Volume in First-Episode Schizophrenia and First-Episode Affective Psychosis: A Cross-Sectional and Longitudinal MRI Study. Biological Psychiatry .Volume 62, Number 7, Pages 773-783. 2007
- T. Vercauteren, X. Pennec, A. Perchant, N. Ayache. Symmetric Log-Domain Diffeomorphic Registration: A Demons-based Approach. MICCAI 2008
- M. Ono, S. Kubik, C.D. Abernathey. Atlas of the Cerebral Sulci. New York: Thieme Medical Publishers. 1990
- Y. Hirayasu, M.E. Shenton, D.F. Salisbury, R.W. McCarley. Hippocampal and superior temporal gyrus volume in first-episode schizophrenia. Arch Gen Psychiatry 2000;57:618–619.